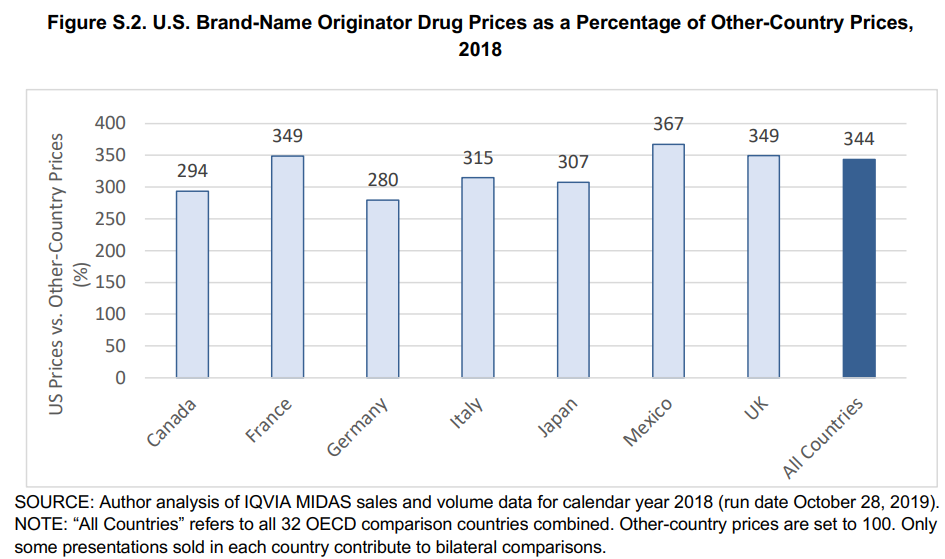
Last week, FDA authorized the State of Florida to import pharmaceuticals from Canada. The price of branded pharmaceuticals in the US is nearly three times the cost of identical pharmaceuticals in Canada, so this approach could save the US money.
While there are some administrative issues to work out on the US side, the biggest issue is the following: Canada doesn’t want to sell. CBC reports that Canadian Health Minister Mark Holland stated:
“There is no way we will allow any jurisdiction, be it a state or another foreign jurisdiction, to endanger the Canadian drug supply. That is not an appropriate solution to whatever challenges they may be facing…
Canadian Health Minister Mark Holland
We’re going to do everything in our power to make sure that another country cannot be given the ability to pillage our health system for its own benefit.”
Canada is concerned because the US would rapidly exhaust the Canadian drug supply. This issue arose in 2019 as well during the Trump administration and Canadians had a similar response back then:
Canada does not have a large enough supply of prescription drugs to meet U.S. demand, and importing medicines from Canada would not significantly lower U.S. prices, Ottawa’s acting ambassador told U.S. officials in recent meetings, according to a statement published on Friday…[the statement] cited a 2019 study that projected that if 40% of U.S. prescriptions were filled from Canada, the Canadian drug supply would run out in 118 days…the U.S. state of Florida spends more on prescription drugs than all of Canada.
https://www.reuters.com/article/us-canada-health-supplies/canadian-ambassador-says-drug-imports-would-not-lower-u-s-prices-idUSKBN1XB55E/
FDA may not take into account that if States begin to import drugs from Canada, pharmaceutical companies may decide to raise prices in Canada closer to US levels, as the US is the largest market for most pharmaceuticals.
Much more to come.